Dimensions
Single Door
Dimensions:
W:77.5 cm
H:180.0 cm
D:105.4 cm
Weight:
382 kg
Double Door
Dimensions:
W:77.5 cm
H:180.0 cm
D:109.5 cm
Weight:
408 kg

Validated for over 23,000 devices from over 100 manufacturers

Terminally sterilizes instruments without toxic residues

Consume approximately 70% less energy per year than steam sterilizers

Saves ∼180,000 liters of water per year compared to steam sterilizers
The STERRAD™ 100NX System with ALLClear™ Technology sterilizes medical devices by diffusing hydrogen peroxide vapor into the chamber and then electromagnetically exciting the hydrogen peroxide molecules into a low-temperature plasma state.
With over 20,000 STERRAD™ Systems installed globally, it is trusted by healthcare institutions around the world to terminally sterilize instruments.
Future Focused System
Upgradeable technology that enhances system capability.
- An expandable, upgradeable sterilization system that enhances processing capability.
- Benefit from the latest technology while preserving the value of your capital investment.
Trusted Sterility
Sterility assurance from the leader in low-temperature sterilization.
- Pioneers in vaporized hydrogen peroxide and hydrogen peroxide gas plasma sterilization technology for over 25 years.
- Over 20,000 medical device manufacturer endorsements.
- The ALLClear™ Technology within the system increases sterilization productivity by conditioning the load, checking for moisture, and running system diagnostics prior to sterilization cycles to maximize successful cycle runs.
Safety Considerations
Designed to minimize healthcare worker chemical exposure.
- Touch-free cassette disposal promotes operators’ safety.1
- The Hydrogen peroxide gas plasma sterilization technology helps enhance the safety of patients and sterile processing staff. Gas plasma is documented to actively break down hydrogen peroxide, reducing emissions exposure for sterile processing staff.2
Reduced Costs
Delivers ongoing economic value.
- STERRAD™ Systems preserve instrument integrity, reducing costly repairs and replacements.3-4
- Reduces annual energy and water consumption per year by ~70% and ~180,000 litres, respectively, compared with autoclave steam sterilizers.5
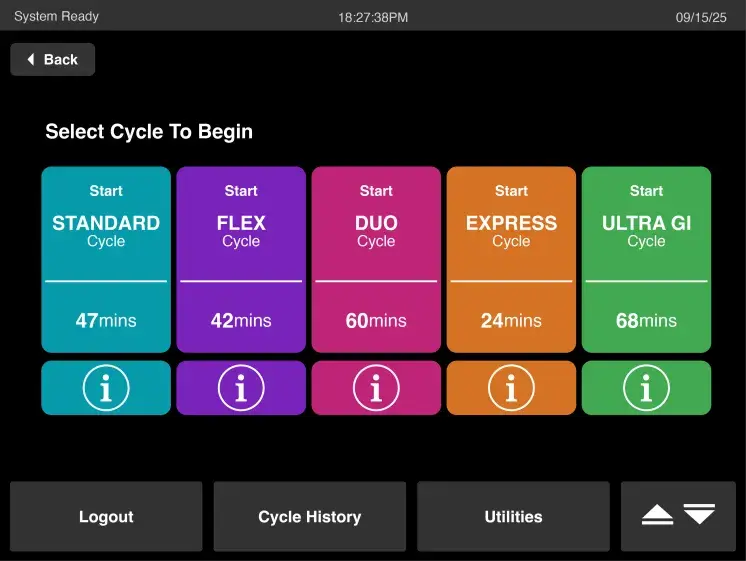
Cycle Information
Includes up to five sterilization cycles – STANDARD, EXPRESS, FLEX, DUO, and ULTRA GI – to sterilize a variety of medical instruments, supported by endorsements from medical device manufacturers.
STANDARD Cycle: 47 Minutes*
- Single Channel Stainless Steel Lumens
- General Medical Instruments
Instruments include, But Are Not Limited To:
- Arthroscope And Laparoscopic Instrument Sets
- Eye Instruments
- Cystoscope Instruments
- Rigid And Semi-rigid Ureteroscopes
- Cameras And Light Cords
- Rechargeable Batteries
- Orthopedic Drills And Saws
- Ultrasound Probes/Transducers
*Cycle times are approximate. ALLClear™ Technology may increase processing time by approximately 5 minutes.
FLEX Cycle: 42 Minutes*
- Single Channel Flexible Endoscopes
- Flexible Endoscopes Without Lumens
Instruments include, But Are Not Limited To:
- Bronchoscopes
- Hysteroscopes
- Cystoscope
- Flexible Ureteroscopes
- Choledochoscopes
- Thoracoscopes
- Intubation Fiberscopes
*Cycle times are approximate. ALLClear™ Technology may increase processing time by approximately 5 minutes.
DUO Cycle: 60 Minutes*
- Single Channel Flexible Endoscopes
- Flexible Endoscopes Without Lumens
- Cameras
- Accessory Light Cords
Instruments include, But Are Not Limited To:
- Bronchoscopes
- Hysteroscopes
- Cystoscope
- Flexible Ureteroscopes
- Choledochoscopes
- Thoracoscopes
- Intubation Fiberscopes
- Light Cords
*Cycle times are approximate.
EXPRESS Cycle : 24 Minutes*
- General Medical Devices Requiring Surface
- Sterilization, Sterilization Of Mated Stainless Steel
- And Titanium Surfaces
Instruments include, But Are Not Limited To:
- da Vinci Endoscopes
- Rigid Or Semi-Rigid Endoscopes Without Lumens
- General Surgery Devices Without Lumens
- Rechargeable Batteries
- Eye Instruments Without Lumens
- Ultrasound Probes/Transducers
*Cycle times are approximate. ALLClear™ Technology may increase processing time by approximately 5 minutes.
ULTRA GI Cycle Processing: 68 Minutes*
ULTRA GI Cycle is designed for sterilization of the following:
- Hydrogen peroxide compatible flexible multi-channel duodenoscopes with no more than 4 channels, with lumen dimensions having an inside diameter (ID) of ≥1mm x ≤1500mm in length, or ≥2mm ID x ≤1630mm in length.
- One flexible duodenoscope per tray, and no more than 2 flexible duodenoscopes per cycle.
Note 1: The STERRAD 100NX Sterilizer ULTRA GI Cycle was validated using a load weight of 15.4 lbs (2 x 7.7 lbs), one endoscope per shelf.
Note 2: Only duodenoscopes that have been cleared as compatible with vaporized hydrogen peroxide are acceptable. Check STERRAD Sterilizer Cycle Selection table for ULTRA GI Cycle compatible duodenoscopes.
*Cycle times are approximate. Integrated ALLClear Technology, included in cycle time
STANDARD Cycle: 47 Minutes*
- Single Channel Stainless Steel Lumens
- General Medical Instruments
Instruments include, But Are Not Limited To:
- Arthroscope And Laparoscopic Instrument Sets
- Eye Instruments
- Cystoscope Instruments
- Rigid And Semi-rigid Ureteroscopes
- Cameras And Light Cords
- Rechargeable Batteries
- Orthopedic Drills And Saws
- Ultrasound Probes/Transducers
*Cycle times are approximate. ALLClear™ Technology may increase processing time by approximately 5 minutes.
FLEX Cycle: 42 Minutes*
- Single Channel Flexible Endoscopes
- Flexible Endoscopes Without Lumens
Instruments include, But Are Not Limited To:
- Bronchoscopes
- Hysteroscopes
- Cystoscope
- Flexible Ureteroscopes
- Choledochoscopes
- Thoracoscopes
- Intubation Fiberscopes
*Cycle times are approximate. ALLClear™ Technology may increase processing time by approximately 5 minutes.
DUO Cycle: 60 Minutes*
- Single Channel Flexible Endoscopes
- Flexible Endoscopes Without Lumens
- Cameras
- Accessory Light Cords
Instruments include, But Are Not Limited To:
- Bronchoscopes
- Hysteroscopes
- Cystoscope
- Flexible Ureteroscopes
- Choledochoscopes
- Thoracoscopes
- Intubation Fiberscopes
- Light Cords
*Cycle times are approximate.
EXPRESS Cycle : 24 Minutes*
- General Medical Devices Requiring Surface
- Sterilization, Sterilization Of Mated Stainless Steel
- And Titanium Surfaces
Instruments include, But Are Not Limited To:
- da Vinci Endoscopes
- Rigid Or Semi-Rigid Endoscopes Without Lumens
- General Surgery Devices Without Lumens
- Rechargeable Batteries
- Eye Instruments Without Lumens
- Ultrasound Probes/Transducers
*Cycle times are approximate. ALLClear™ Technology may increase processing time by approximately 5 minutes.
ULTRA GI Cycle Processing: 68 Minutes*
a class="text-primary" href="https://www.asp.com/en-gb/products/terminal-sterilization/ultra-gi">ULTRA GI Cycle is designed for sterilization of the following:
- Hydrogen peroxide compatible flexible multi-channel duodenoscopes with up to 4 or 8 channels, with lumen dimensions having an inside diameter (ID) of ≥1 mm × ≤1500 mm length, or ≥2 mm × ≤1662 mm length.
- One flexible duodenoscope per tray, and no more than 2 flexible duodenoscopes per cycle.
Note 1: The STERRAD 100NX Sterilizer ULTRA GI Cycle was validated using a load weight of 6.98 kg (0.90 x 3.49 kg) , one endoscope per shelf.
Note 2: Only duodenoscopes that have been cleared as compatible with vaporized hydrogen peroxide are acceptable. Check STERRAD Sterilizer Cycle Selection table for ULTRA GI Cycle compatible duodenoscopes.
*Cycle times are approximate. Integrated ALLClear Technology, included in cycle time
IFU
The ASP IFU and User Guide library can be used to search for the appropriate IFU, User Guides and Data Sheets for ASP products. Product literature and availability varies by country; users can ensure they locate the correct literature for their setting by searching by product and country. Use the link below to locate the STERRAD™ 100NX IFU.
Device Validations
Have questions about your STERRAD™ System?
Find up-to-date compatibility information and helpful resources in the online STERRAD™ Sterility Guide.
Education and Training
ASP Learning Lab is a dedicated resource platform for healthcare professionals. Access factsheets, webinars, whitepapers, and technical resources on low-temperature sterilization, medical device reprocessing, validation standards, and industry best practices, all designed to support your infection prevention expertise.
References
- Advanced Sterilization Products. STERRAD™ 100NX System with ALLClear™ Technology Data Sheet.AD-160028-01-CT_D-MDR.
- Advanced Sterilization Products. STERRAD™ Systems with ALLClear™ Technology Product Brochure. AD-160029-01-CT_D-MDR.
- Advanced Sterilization Products. STERRAD™ Superiority Factsheet Volume 1. The Power of Plasma. AD-200071-01-CT_B-MDR.
- Advanced Sterilization Products. STERRAD™ Superiority Factsheet Volume 4. The Importance of Instrument Validation. AD-200071-01-CT_B-MDR.
- Advanced Sterilization Products. STERRAD NX™ with ALLClear™ Technology User Guide.
Advanced Sterilization Products. STERRAD VELOCITY™ Biological Indicator System Product Brochure. AD-170021-01-CT_C-MDR. - Advanced Sterilization Products. STERRAD™ 100NX System with ALLClear™ Technology User Guide.
- Advanced Sterilization Products. Comparison Study of Environmental Hydrogen Peroxide Levels of STERRAD™ Systems and STERIS V-PRO™ Low Temperature Sterilizers Reveals Striking Differences. 2016.
- Advanced Sterilization Products. Assessment of Operating Costs Due to Energy and Water Use During Terminal Sterilization With STERRAD™ Systems Compared to a Steam Sterilizer. AD-160024-01-CT_C-MDR. 2016.
- Advanced Sterilization Products. ASP ACCESS™ Technology Product Brochure. AD-160030-01-CT_C-MDR.
Advanced Sterilization Products. ASP Ecosystem Brochure - Innovation to Advance the Quality of Patient Care. AD-170035-01-CT_D-MDR.